UF researchers studying the use of a noninvasive brain stimulation treatment paired with cognitive training have found the therapy holds promise as an effective, drug-free approach for someday warding off Alzheimer’s disease and other dementias.
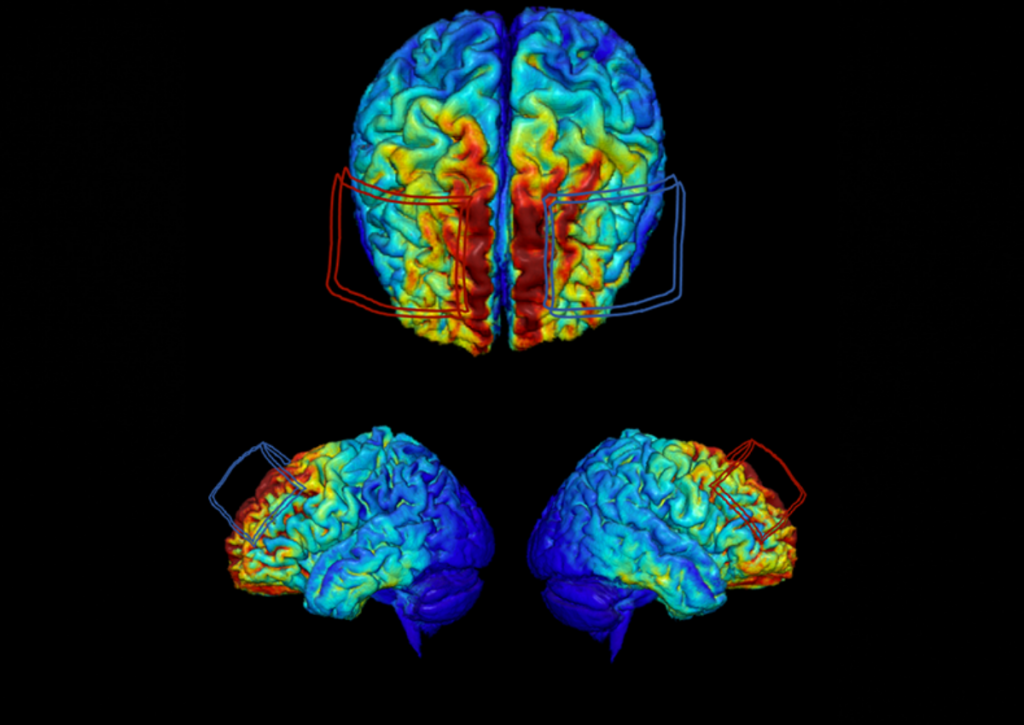
Yet determining optimal dosing for the treatment known as transcranial direct current stimulation, or tDCS, which is delivered by a safe and weak electrical current passed through electrodes placed on a person’s head, has been a challenge because of individual differences in anatomy.
Now, with the support of a $2.9 million grant from the National Institute on Aging, UF scientists will use artificial intelligence technology—including advanced machine learning and deep-learning algorithms—to evaluate a large data set gleaned from a study of tDCS and cognitive training in older adults. The goal is to gain more insight into the mechanisms driving treatment response and individual variability so that researchers can design a customized method for providing tDCS with the best possible outcome.
“We are excited to evaluate how artificial intelligence can help us understand individual differences in treatment response and build the first-of-its-kind precision dosing system in tDCS augmented cognitive training,” said Dr. Ruogu Fang, one of the study’s principal investigators and an assistant professor of biomedical engineering at the UF College of Engineering. “We hope this system will pave the path for preventing cognitive decline in older adults and changing the trajectory toward Alzheimer’s disease. This framework of AI can be generalized to a variety of brain stimulation methods and neurodegenerative diseases.”
The new study will use data derived from the National Institute on Aging-funded Augmenting Clinical Training in Older Adults: The ACT Study, an ongoing study of 360 older adults led by Dr. Adam Woods, an associate professor of clinical and health psychology in the UF College of Public Health and Health Professions. The ACT study pairs tDCS with cognitive training designed to improve working memory and processing speed.
In a smaller pilot study of the treatment, Woods and his colleagues demonstrated that tDCS combined with cognitive training improved cognition and brain function after only two weeks. Treatment effects varied from person to person due to differences in anatomy, such as skull thickness.
“In the field of tDCS, a fixed dosing approach is the standard convention,” said Woods, also a principal investigator on the new AI study. “All people receive exactly the same dose of tDCS, even though we know that individual differences in head and brain anatomy significantly alter how much current enters the brain and where it goes. Well, it turns out where the current goes and how much enters brain tissue are precisely the features that best predict whether someone will respond to tDCS treatment.”
The new study will leverage machine learning algorithms to evaluate data from behavioral outcomes, neuroimaging and physiological features captured from participants in the ACT study. That will help researchers find patterns among the 16 million data points, said team member Dr. Aprinda Indahlastari, a research assistant professor of clinical and health psychology.
“Once we determine the important factors that drive successful tDCS application, we can strategize how to improve tDCS application by personalizing the treatment dose to each individual older adult,” Indahlastari said. “We can potentially prevent dementia by applying effective intervention methods, such as tDCS, that are tailored to everyone.”
Team member Alejandro Albizu, a doctoral student in neuroscience at the UF College of Medicine, said the study will provide an opportunity to break new ground: “This project will enable us to generate one of the first personalized medicine approaches to combat age-related cognitive decline.”
Suggested Articles
No related articles found.



