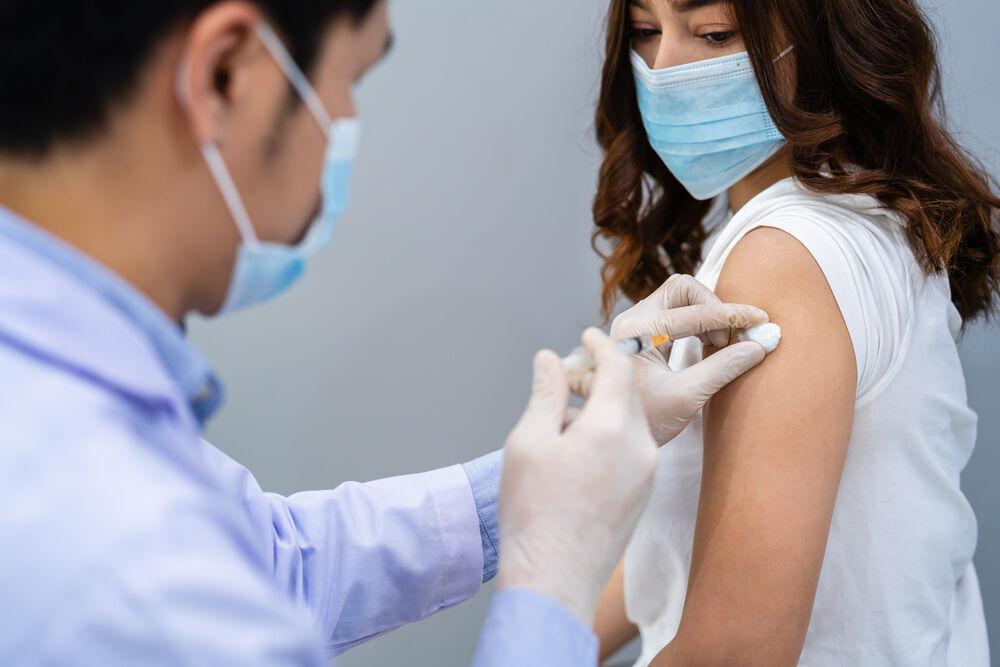
Adolescents could have a second coronavirus vaccine option in the near future. Moderna said Tuesday its shots appeared 93 percent effective at protecting kids as young as 12 two weeks after the first dose.
When will the vaccines be available? Earlier this month, U.S. and Canadian health officials authorized the Pfizer two-dose vaccine for children aged 12 and up. Moderna says it will submit its teen data to the FDA next month.
The company studied more than 3,700 kids, aged 12 to 17. Early findings suggest the vaccine triggers the same signs of immune protection in kids as it does in adults, as well as the same kind of temporary side effects like sore arms, headache, and fatigue.
Both Pfizer and Moderna have begun testing smaller doses in even younger children, from age 11 down to 6-month-old babies.
This story originally appeared in WORLD. © 2021, reprinted with permission. All rights reserved.
Suggested Articles
No related articles found.



