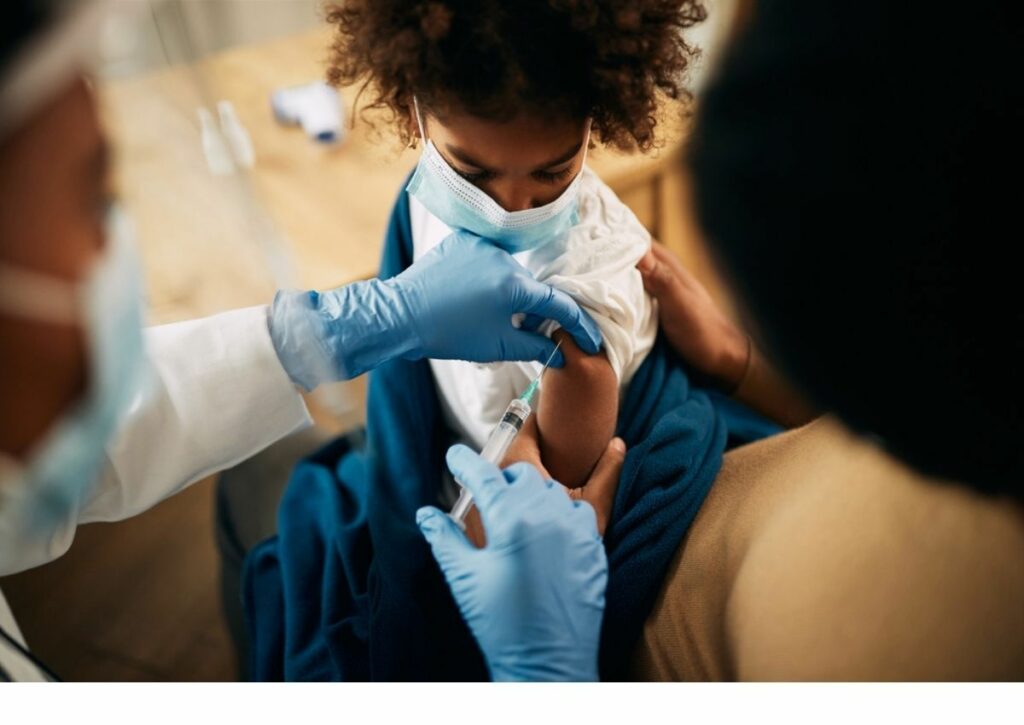
Moderna announced Monday that it will submit interim data to federal regulators to support its bid to become the next COVID-19 vaccine authorized for emergency use in children.
The study gave two half-doses of the vaccine to participants ages 6 through 11. Moderna reported that the children who received the vaccine had antibody levels 1.5 times higher than those of adults who were vaccinated.
A similar application from Pfizer is already under U.S. Food and Drug Administration review. If approved soon, children could start getting vaccinated in early November.
The FDA still has not made a decision on Moderna’s application to give its vaccine to teenagers.
Is it safe? The study of 4,753 participants was too small to draw conclusions on rare side effects such as heart inflammation, and it has not been submitted to a scientific journal for peer review.
Moderna reported the same temporary side effects adults have experienced, including fatigue, headaches, low-grade fevers, and soreness. Researchers will continue to monitor the children involved for a year.
Moderna is also setting up trials to vaccinate even younger children. It said it has already enrolled 5,700 kids between 6 months and 5 years old in the United States and Canada and is still accepting applications.
This story originally appeared in WORLD. © 2021, reprinted with permission. All rights reserved.
Suggested Articles
No related articles found.



