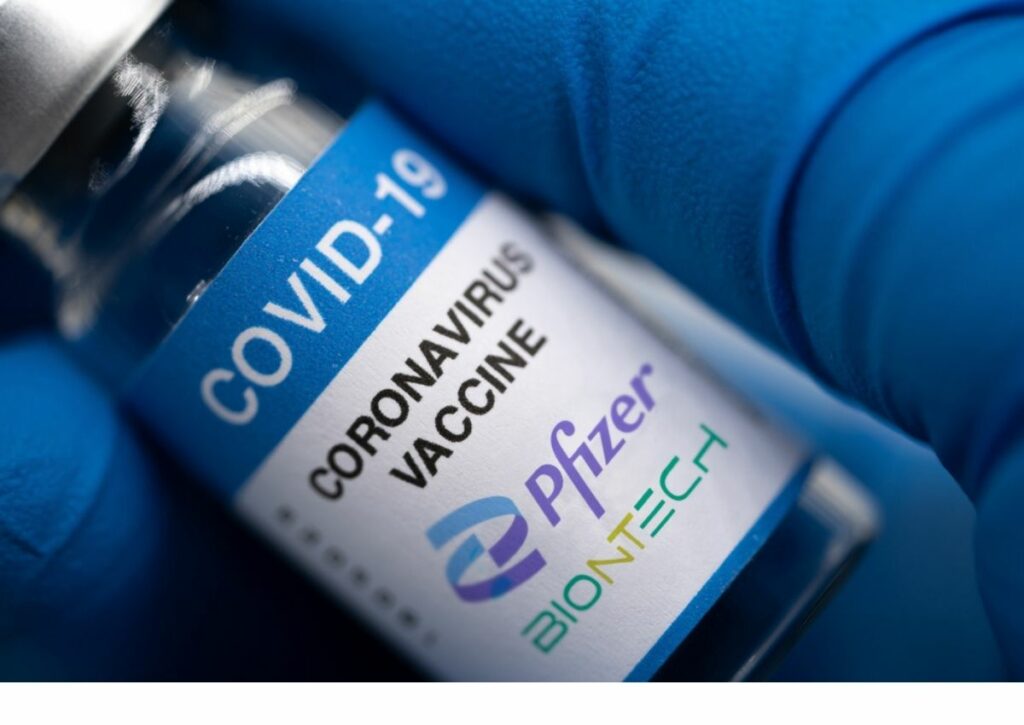
Moderna and Pfizer-BioNTech received approval Monday from the Food and Drug Administration to update their COVID-19 shots to combat more variant strands of the virus.
The new formula specifically targets the omicron subvariant XBB.1.5. The FDA gave the vaccines full approval for people ages 12 and up, and it granted emergency use authorization for children six months to 11 years old.
On Tuesday, the Centers for Disease Control and Prevention added its recommendation to the FDA’s approval and authorization.
How widespread is COVID-19 vaccination these days? Only about 20% of U.S. adults got the most recent coronavirus booster shot that was offered last year. The number of new COVID-19 hospitalizations rose over the summer but remained less than half of last summer’s peak of about 46,000.
Since the federal government ended the pandemic public health emergency in May, the CDC no longer tracks the number of positive test results.
This story originally appeared in WORLD. © 2023, reprinted with permission. All rights reserved.
Suggested Articles
No related articles found.



